

Molecules of the same type follow this trend. While the molecular weight is the sum of the atomic weights of the elements present in the molecule.įor elements, the greater the molar mass, the greater the boiling point. The molecular mass is the sum of the atomic masses of the elements present in the molecule. 1 mole 6.022 x 10 23 units of whatever it is you’re measuring. A typical chemical reaction involves more atoms and molecules than is feasible to count, hence chemists use moles to represent them. PubChem is a reliable source of chemical information from authoritative sources. Before we delve into molarity and molality, we need to understand why moles matter in chemistry.

You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. It is calculated by adding the number of protons and neutrons (electrons are considered too light and are negligible). PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. The atomic mass is the mass of a single atom/isotope. The atomic weight of an element is an average of the weight of its isotopes with respect to their relative abundances. The molecular mass would then be: $$(2*1.01\frac$$ Water is made up of 2 hydrogens and 1 oxygen, and the atomic mass of hydrogen is 1.01 g/mol and the atomic mass of oxygen is 16.00 g/mol. The molecular weight of water can be calculated by adding together the atomic masses of the elements in it. Thus, the context and questions must be clear when discussing these terms! Always be careful when reading textbooks and problems to see if when "molecular mass" is mentioned they actually mean "molecular mass" or if they mean "molecular weight" instead.Īs a guide to get you used to this, if I am using a term with its original definition, it will be in green, if I am using it with its colloquial definition it will be in red. Pretend you have 400 g of NaCl in 1.5 L of H 2 O. The Mole How are chemists able to measure things when everything is made up of different atoms and, to make this more difficult, different isotopes of those atoms? With moles, of course! Not with the small subterranean mammal (though they are cute), but the unit moles.Īs I mentioned before, atomic mass and atomic weight are often used interchangeably, so molar mass, molecular mass, AND molecular weight are also often used interchangeably. If you dont know the moles but you have the mass, you can still figure out the molarity by using the mole ratio. Lastly, we will look at the link between molar mass and boiling point.
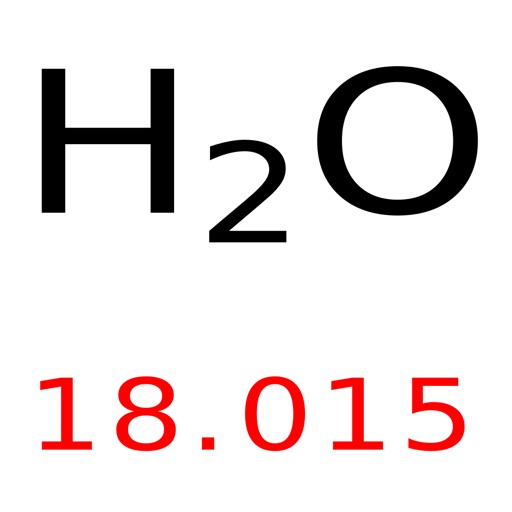
Then, we will explain the concept of molecular mass and explain the differences between it and molar mass.Next, we will explain the concepts of atomic mass and molar mass and the differences between the two.First, we will define a mole and explain why that unit is used.This article is about moles and molar mass.In this article, we will discuss how chemists measure and group the masses of atoms and molecules. What is the mole fraction of ethanol in the. suppose you have a solution made of 300 g of ethanol and 500 g of water. In both cases, it is the mass of 6.02 × 1023 molecules. One mole of carbon dioxide molecules has a mass of 44.01g. what is the molarity of a solution prepared by dissolving 8 g of BaCL2 in enough water to make 450.0 mL solution 0.085 M. The molar mass of any compound is the mass in grams of one mole of that compound. However, we first need to know the average mass of each atom. molarity and mole fraction measure whichof the following properties concentration of a solution. We treat atoms the same way! To find out how many atoms are in a sample, we can relate the mass of the sample to the number of atoms. If you know the mass, you can multiply that by the desired quantity and add the candy to a scale until you reach that weight. The difference is that molecular mass is the mass of one specific particle or molecule, while the molar mass is an average over many particles or molecules. This is a very long and tedious way to do business! Instead, you utilize the average mass of the candy. (credit: modification of work by Mark Ott)Ī simple mathematical relationship can be used to relate the volumes and concentrations of a solution before and after the dilution process.Let's say you work at a candy store and a customer asks for different amounts of candy (100, 250, 1000…), so you count out each order. Explore molarity definition, uses, and applications in chemistry at BYJUS. \): A solution of \(KMnO_4\) is prepared by mixing water with 4.74 g of KMnO4 in a flask. Molarity And Mole Fraction - Get introduced to the concepts of molarity, molality, mole fraction, mass percent/weight percent and other related concepts.
